Precipitate chemistry deff11/11/2023  Precipitation reactions are also useful to extract elements, like magnesium, from seawater. For example, a barium nitrate solution will react with sulfate ions to form a solid barium sulfate precipitate, indicating that it is expected that sulfate ions are present. Precipitate development is valuable in the detection of the type of cation in a salt. Subsequently, the precipitate may easily be separated by filtration/decanting or centrifugation. Precipitation chemistry def Precipitation weather Britannica Precipitate Definition & Meaning Coprecipitation - Wikipedia Separation by. DMA and DEF were useful as, depending on whether a solvent binding to the. Antisolvent is added - This radically drops the solubility of the desired product. a solvent and heating to evaporate some solvent and precipitate a crystal.Using a supersaturated solution - Without sufficient force of gravity (settling) to bring the solid particles together, the precipitate remains in suspension.The chemical that leads the solid to form is called the precipitant. Temperature of a solution is lowered - The lower temperature reduces the solubility of a salt, resulting in its precipitation as a solid.Two soluble salts react in solution to form one or more insoluble products.Powders derived from precipitation are known as flowers. The precipitate-free liquid remaining above the solid is called the supernate or supernatant. A precipitate will form if a solution containing one of these anions is added to a solution containing a metal cation such as Fe 2+, Cu 2+, or Al 3+. Compounds having anions such as sulfide (S 2 ), hydroxide (OH ), carbonate (CO 32 ), and phosphate (PO 43) are often insoluble in water.  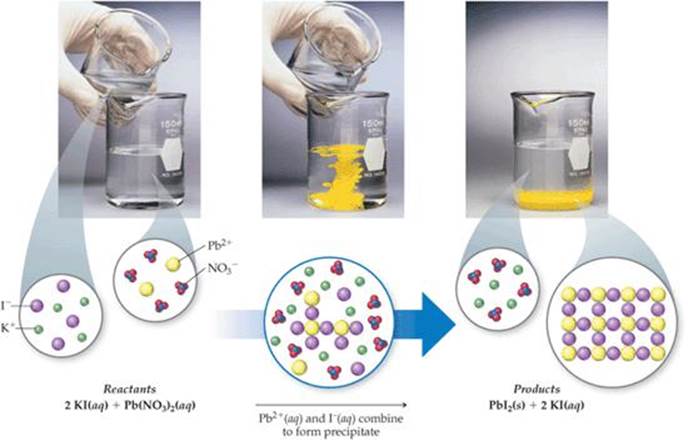
Precipitate is referred to as a pellet after sedimentation when using a centrifuge to press it into a compact mass. The solid that separates is called a precipitate. When the reaction occurs in a liquid solution, the solid formed is called the precipitate. 
Both compounds are white solids that can be dissolved in water to make clear, colorless solutions. One example is the reaction between lead (II) nitrate and potassium iodide. Precipitation is the creation of a solid in a solution or inside another solid during a chemical reaction or by diffusion in a solid. A precipitation reaction is when two aqueous ionic compounds form a new ionic compound that is not soluble in water. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
